General Principles and Processes of Isolation of Elements (Not for Exams)-Solutions
CBSE Class 12 Chemistry
NCERT Solutions
Chapter - 06
General Principles and Processes of Isolation of Elements
In-text question
1. Which of the ores mentioned in Table 6.1 can be concentrated by magnetic separation method?
Ans. If the ore or the gangue particles which are magnetic in nature can be attracted by the magnetic field, then the ore can be concentrated by the process of magnetic separation. Among the ores mentioned in table 6.1, the ores of iron such as haematite (Fe2O3), magnetite (Fe3O4), siderite (FeCO3) and iron pyrites (FeS2)can be separated by the process of magnetic separation.
2. What is the significance of leaching in the extraction of aluminium?
Ans.Leaching with a concentrated solution of NaOH helps to remove the impurities of iron oxide and TiO2 from bauxite. In the extraction of aluminium, the significance of leaching is to concentrate pure alumina (Al2O3)from bauxite ore. Bauxite usually contains silica, iron oxide, and titanium oxide as impurities. In the process of leaching, alumina is concentrated by digesting the powdered ore with a concentrated solution of NaOH at 473-523 K and 35-36 bar. Under these conditions, alumina (Al2O3) dissolves as sodium meta-aluminate and silica (SiO2) dissolves as sodium silicate leaving the impurities behind.
The impurities are then filtered and the solution is neutralized by passing CO2 gas. In this process, hydrated (Al2O3)gets precipitated and sodium silicate remains in the solution. Precipitation is induced by seeding the solution with freshly prepared samples of hydrated (Al2O3).
Hydrated alumina thus obtained is filtered, dried, and heated to give back pure alumina (Al2O3).
3. The reaction, is thermodynamically feasible as is apparent from the Gibbs energy value. Why does it not take place at room temperature?
Ans. The change in Gibbs energy is related to the equilibrium constant, K as = -RT ln K. Though the reaction is feasible but it does not takes place at room temperature since all the reactants and the products at room temperature are solids. However, at higher temperature, when chromium melts, the reaction proceeds rapidly.
We also know that according to the equation . Increasing the temperature increases the value of making the value of 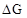 more and more negative. Therefore, the reaction becomes more and more feasible as the temperature is increased.
more and more negative. Therefore, the reaction becomes more and more feasible as the temperature is increased.
4. Is it true that under conditions, Mg can reduce SiO2 and Si can reduce MgO? What are those conditions
Ans.
The temperature range in which is more negative than , Mg can reduce SiO2 to Si.
.
On the other hand, the temperature range in which is more negative than , Si can reduce MgO to Mg..The temperature at which curves of these two substances intersect is 1966 K. Thus, below 1966 K, Mg can reduce SiO2 but above 1966 K, Si can reduce MgO.
Chapter End Question
1. Copper can be extracted by hydrometallurgy but not zinc. Explain.
Ans. The reduction potentials of zinc (Zn 2+/Zn= -0.76 V) and iron (Fe 2+/Fe = -0.44 V) are lower than that of copper (Cu 2+/Cu= +0.34 V). This means that zinc is stronger reducing agent than copper and easily displace Cu from the solution of Cu2+ ions. In hydrometallurgy, zinc and iron can be used to displace copper from their solution. Zn(s) + Cu2+(aq) Zn2+(aq) + Cu(s)
On the bother hands, to displace zinc from the solution of Zn2+, we need a stronger reducing agent like Mg, Ca, K, etc. But all these metals react with water with the evolution of H2 gas. . As a result, these metals cannot be used in hydrometallurgy to extract zinc.Hence, copper can be extracted by hydrometallurgy, but not zinc.
2. What is the role of depressant in froth floatation process?
Ans.It is a substance which suppresses or depresses the tendency of a particular metal sulphide to form froth in the froth floatation process by combining with it. For example, to separate two sulphide ores (ZnS and Pbs), NaCN is used as a depressant which selectively allows PbS to come with froth, but prevents ZnS from coming to froth. This happens because NaCN reacts with ZnS to form Na2[Zn(CN)4].
4NaCN + ZnS Na2[Zn(CN)4] + Na2S.
Under these conditions , only PbS forms froth and hence can be separated from ZnS ore.
3. Why is the extraction of copper from pyrites more difficult than that from its oxide ore through reduction?
Ans. The standard free energy of Cu2S is more than those of H2S and CS2. Therefore, H2 and C cannot reduce Cu2S to Cu.These reactions are not feasible.However, the Gibbs free energy of Cu2O is lower than that of CO. Hence, C can reduce Cu2O to Cu.
Hence, the extraction of copper from its pyrite ore is difficult than from its oxide ore through reduction.
4. Explain: (i) Zone refining (ii) Column chromatography.
Ans. (i) Zone refining: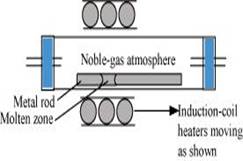
This method is based on the principle that impurities are more soluble in the molten state of metal (the melt) than in the solid state. In the process of zone refining, a circular mobile heater is fixed at one end of a rod of impure metal. As the heater moves, the molten zone of the rod also moves with it. As a result, pure metal crystallizes out of the melt and the impurities pass onto the adjacent molten zone. As the heater is slowly moved along the length of the rod, the pure metal crystallises out of the melt whereas the impurities pass into the adjacent molten zone. This process is repeated several times, till the impurities are completely driven to one end of the rod which is then cut off and discarded. The process is usually carried out in an inert atmosphere to prevent the oxidation of the metal. Silicon, boron, gallium, indium etc. can be purified by this process.
(ii) Column chromatography:
Column chromatography is a technique used to separate different components of a mixture. It is a very useful technique used for the purification of elements available in minute quantities. It is also used to remove the impurities that are not very different in chemical properties from the element to be purified. Chromatography is based on the principle that different components of a mixture are differently adsorbed on an adsorbent. In chromatography, there are two phases: mobile phase and stationary phase. The stationary phase is immobile and immiscible. Al2O3 column is usually used as the stationary phase in column chromatography. The mobile phase may be a gas, liquid, or supercritical fluid in which the sample extract is dissolved. Then, the mobile phase is forced to move through the stationary phase. The component that is more strongly adsorbed on the column takes a longer time to travel through it than the component that is weakly adsorbed. The adsorbed components are then removed (eluted) using a suitable solvent (eluant).
5. Out of C and CO, which is a better reducing agent at 673 K?
Ans. From the Ellingham diagram, we find that at 983K, the curves intersect.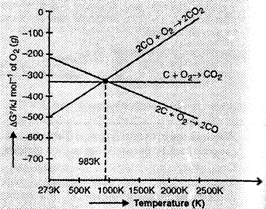
The value of for change of C to CO2 is less than the value of for change of CO to CO2. Therefore, coke (C) is better reducing agent than CO at 983K or above temperature. However, below this temperature (e.g. 673K). CO is more effective reducing agent than C.
6. Name the common elements present in the anode mud in electrolytic refining of copper. Why are they so present?
Ans. In electrolytic refining of copper, the common elements present in anode mud are selenium, tellurium, silver, gold, platinum, and antimony.
These elements are very less reactive and are not affected during the purification process. Hence, they settle down below the anode as anode mud.
7. Write down the reactions taking place in different zones in the blast furnace during the extraction of iron.
Ans. During the extraction of iron, the reduction of iron oxides takes place in the blast furnace. In this process, hot air is blown from the bottom of the furnace and coke is burnt to raise the temperature up to 2200 K in the lower portion itself. The temperature is lower in the upper part. Thus, it is the lower part where the reduction of iron oxides (Fe2O3 and Fe3O4) takes place.
(a) Zone of reduction or top zone (823K) :The reactions taking place in the lower temperature range (500 - 800 K) in the blast furnace are:
(b) Zone of slag formation: The silicate impurities of the ore is removed as slag by calcium oxide (CaO), which is formed by the decomposition of limestone CaCO3.
(c) Zone of fusion: The reaction taking place in the higher temperature range (900 – 1500 K) in the blast furnace are:
(d) Zone of combustion (2170 K) : Carbon combines with oxygen to form carbon dioxide: C+ O2 CO2 ; 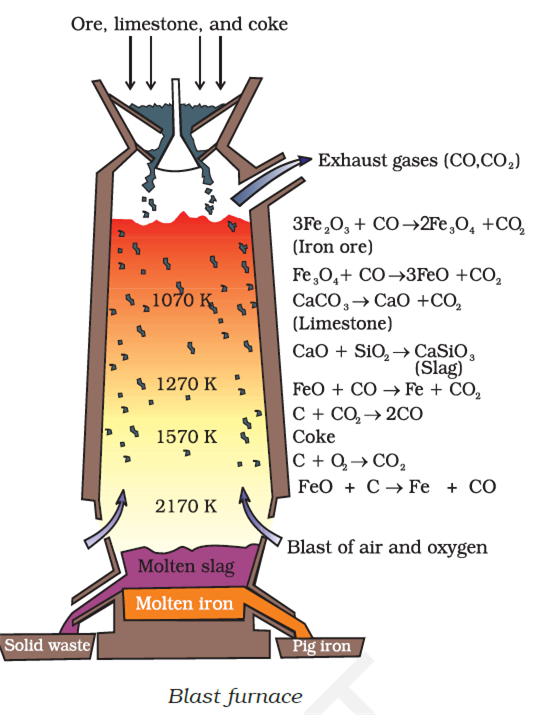
8. Write chemical reactions taking place in the extraction of zinc from zinc blende.
Ans. The different steps involved in the extraction of zinc from zinc blende (ZnS) are given below:
(i) Concentration of ore First, the gangue from zinc blende is removed by the froth floatation method.
(ii) Conversion to oxide (Roasting): Sulphide ore is converted into oxide by the process of roasting. In this process, ZnS is heated in a regular supply of air in a furnace at a temperature, which is below the melting point of Zn.
(iii) Extraction of zinc from zinc oxide (Reduction): Zinc is extracted from zinc oxide by the process of reduction. The reduction of zinc oxide is carried out by mixing it with powdered coke and then, heating it at 673 K.
(iv) Electrolytic Refining: Zinc can be refined by the process of electrolytic refining. In this process, impure zinc is made the anode while a pure zinc strip is made the cathode. The electrolyte used is an acidified solution of zinc sulphate (ZnSO4). Electrolysis results in the transfer of zinc in pure from the anode to the cathode.
Anode: Zn Zn2+ + 2e-
Cathode: Zn2+ + 2e- Zn
9. State the role of silica in the metallurgy of copper.
Ans. Silica (SiO2) acts as an acidic flux in the metallurgy of copper. it helps in the removal of basic impurities, FeO as iron silicate (FeSiO3) slag, while copper is produced in the form of copper matte. During the roasting of pyrite ore, a mixture of FeO and Cu2O is obtained.
10. What is meant by the term "chromatography"?
Ans. Chromatography is a collective term used for a family of laboratory techniques for the separation of mixtures. The term is derived from Greek words 'chroma' meaning 'colour' and 'graphic' meaning 'to write'. Chromatographic techniques are based on the principle that different components are absorbed differently on an absorbent. Initially, chromatography was used to identify and separate only the coloured components or constituents. But, now any type of constituent can be separated by this method. There are several chromatographic techniques such as paper chromatography, column chromatography, gas chromatography, etc.
11. What criterion is followed for the selection of the stationary phase in chromatography?
Ans. The stationary phase is selected in such a way that the components of the sample have different solubility's in the phase. Hence, different components have different rates of movement through the stationary phase and as a result, can be separated from each other.
In chromatography particularly in adsorption chromatography, the stationary phase is the adsorbent. It should fulfill the following criteria for better results.
(a) It should have high but selective adsorption power.
(b) That stationary phase is selected in which impurities are most strongly adsorbed than the element to be purified.
(c) The adsorbent should not react chemically with the solvents used for elution or with the components of the mixture under investigation.
(d) The adsorbent should be perfectly white.
12. Describe a method for refining nickel.
Ans. Nickel is refined by Mond's process. In this process, nickel is heated in the presence of carbon monoxide to form nickel tetracarbonyl, which is a volatile complex.
Then, the obtained nickel tetracarbonyl is decomposed by subjecting it to a higher temperature (450 - 470 K) to obtain pure nickel metal.
13. How can you separate alumina from silica in bauxite ore associated with silica? Give equations, if any.
Ans. To separate alumina from silica in bauxite ore associated with silica, first the powdered ore is digested with a concentrated NaOH solution at 473 - 523 K and 35 - 36 bar pressure. This results in the leaching out of alumina (Al2O3) as sodium aluminate and silica (SiO2) as sodium silicate leaving the impurities behind.
Then, CO2 gas is passed through the resulting solution to neutralize the aluminate in the solution, which results in the precipitation of hydrated alumina. To induce precipitation, the solution is seeded with freshly prepared samples of hydrated alumina.
During this process, sodium silicate remains in the solution. The obtained hydrated alumina is filtered, dried, and heated to get back pure alumina.
14. Giving examples, differentiate between 'roasting' and 'calcination'.
Ans.
| S.No. | Roasting | Calcination |
| 1. | Roasting is the process of converting sulphide ores to oxides by heating the ores in a regular supply of air at a temperature below the melting point of the metal. | calcination is the process of converting hydroxide and carbonate ores to oxides by heating the ores either in the absence or in a limited supply of air at a temperature below the melting point of the metal. |
| 2. | For example, sulphide ores of Zn, Pb, and Cu are converted to their respective oxides by this process. | For example, hydroxide of Fe, carbonates of Zn, Ca, Mg are converted to their respective oxides by this process. |
Chapter End Question
15. How is 'cast iron' different from 'pig iron"?
Ans. Pig Iron: The iron obtained from blast furnaces is known as pig iron. It contains around 4% carbon and many impurities such as S, P, Si, Mn in smaller amounts.
Cast Iron: Cast iron is obtained by melting pig iron and coke using a hot air blast. It contains a lower amount of carbon (3%) than pig iron. Unlike pig iron, cast iron is extremely hard and brittle.
16. Differentiate between "minerals" and "ores".
Ans. Both ores and minerals are the compounds of metal occurring in the earth's crust but from the ore the metal can be extracted profitably and conveniently. The extraction of metal from the mineral can be very tedius , for example Bauxite and clay the two minerals of aluminum occurring in the earth's crust. Out of these two minerals, bauxite is the ore of aluminium because Al can be extracted profitably and conveniently from bauxite. similarly, there are many minerals containing zinc, but zinc cannot be extracted profitably (conveniently and economically) from all these minerals. Zinc can be obtained from zinc blende (ZnS), calamine ZnCO3, Zincite (ZnO) etc. Thus, these minerals are called ores of zinc. So, minerals from which metal can be profitably and conveniently extracted are known as Ore.
17. Why copper matte is put in silica lined converter?
Ans. Copper matte contains Cu2S and FeS. Copper matte is put in a silica-lined converter to remove the remaining FeO and FeS present in the matte as slag FeSiO3. Also, some silica is added to the silica-lined converter. Then, a hot air blast is blown. As a result, the remaining FeS and FeO are converted to iron silicate FeSiO3 and Cu2S is converted into metallic copper.
Some of the Cu2S undergoes oxidation to form Cu2O which is then reduced to Cu metal.
Thus, copper matte is heated in silica lined converter to remove FeS present in matte as FeSiO3 slag.
18. What is the role of cryolite in the metallurgy of aluminium?
Ans. Cryolite (Na3AlF6) has two roles in the metallurgy of aluminium:
1. To decrease the melting point of the mixture from 2323 K to 1140 K.
2. To increase the electrical conductivity of Al2O3.
19. How is leaching carried out in case of low grade copper ores?
Ans.From the low grade copper (in which the percentage of the metal is quite less), copper is extracted by hydro metallurgy. In this ore is leached out by a suitable acid or bacteria when Cu2+ ions go into the solution. These are then reduced by scarp iron or by passing H2 gas to the metallic form.
The resulting solution is treated with scrap iron or H2 to get metallic copper.
20. Why is zinc not extracted from zinc oxide through reduction using CO?
Ans. The chemical reaction involving in the reduction of ZnO by CO is not feasible thermodnamically. The standard Gibbs free energy of formation of ZnO from Zn is lower than that of CO2 from CO. Therefore, CO cannot reduce ZnO to Zn. Hence, Zn is not extracted from ZnO through reduction using CO.
21. The value of for formation of Cr2O3 is -540 KJmol-1 and that of Al2O3 is -827 kJmol-1. Is the reduction of Cr2O3 possible with Al?
Ans. The value of for the formation of Cr2O3 from Cr (-540 KJmol-1) is less negative than that of Al2O3 from Al (-827 kJmol-1). Therefore, Al can reduce Cr2O3 to Cr. Hence, the reduction of Cr2O3 with Al is possible.
Alternatively,
2Al + ; =-827 kJmol-1
;=-540 KJmol-1
Subtracting equation (ii) from (i), we have
; = - 827 -(-540)= -287 KJmol-1
As for the reduction reaction of Cr2O3 by Al is negative, this reaction is possible.
22. Out of C and CO, which is a better reducing agent for ZnO?
Ans. .......(1)
..........(2)
In first case, there is increase in the magnitude of while in the second case, it almost remains same.
In other words, Reduction of ZnO to Zn is usually carried out at 1673 K. From the above figure, it can be observed that above 1073 K, the Gibbs free energy of formation of CO from C and above 1273 K, the Gibbs free energy of formation of CO2 from C is more negative than the Gibbs free energy of formation of ZnO above. Therefore these particular temperatures.SO, C can easily reduce ZnO to Zn. On the other hand, the Gibbs free energy of formation of CO2 from CO is always less negative than the Gibbs free energy of formation of ZnO. Therefore, CO cannot reduce ZnO. Hence, C is a better reducing agent than CO for reducing ZnO.
23. The choice of a reducing agent in a particular case depends on thermodynamic factor. How far do you agree with this statement? Support your opinion with two examples.
Ans. Thermodynamic factor has a major role in selecting the reducing agent for a particular reaction. Only that reagent will be preferred which will lead to decrease in the free energy at a certain specfic temperature e.g. can reduce ZnO to Zn but not CO. The two reduction reactions are :
............(1)
........(2)
In first case, there is increase in the magnitude of while in the second case, it almost remains same. Also from Ellingham diagram:
The above figure is a plot of Gibbs energy vs. T for formation of some oxides. It can be observed from the above graph that a metal can reduce the oxide of other metals, if the standard free energy of formation of the oxide of the former is more negative than the latter. For example, since is more negative than , Al can reduce Cu2O to Cu, but Cu cannot reduce Al2O3. Similarly, Mg can reduce ZnO to Zn, but Zn cannot reduce MgO because is more negative than .
24. Name the processes from which chlorine is obtained as a by-product. What will happen if an aqueous solution of NaCl is subjected to electrolysis?
Ans. Chlorine is obtained as the by-product in the following processes:
(a) Manufacture of sodium by Down's process.
(b) Manufacture of sodium hydroxide or caustic soda in Nelson's cell.
In Down's Process, molten NaCl is subjected to electrolysis. During electrolysis, Sodium is obtained at cathode while Cl2 is obtained at the anode as a by product.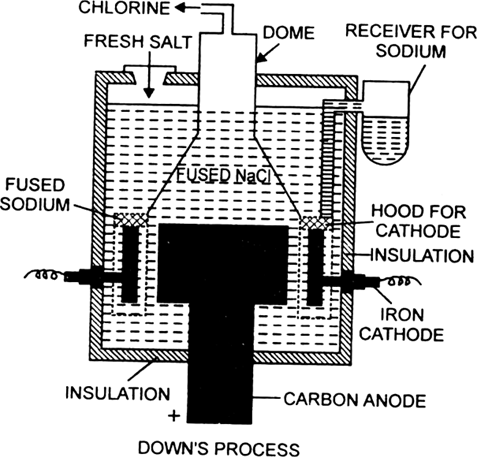
NaCl(melt) Na+(melt) + Cl-(melt)
At cathode: Na+(melt) + e-Na(s)
At anode: Cl-(melt)Cl(g) + e-
2Cl(g) Cl2(g)
The overall reaction is as follows:
If an aqueous solution of NaCl is electrolyzed, Cl2 will be obtained at the anode but at the cathode, H2 will be obtained (instead of Na). This is because the standard reduction potential of Na (E°=-2.71 V) is more negative than that of H2O (E°=-0.83 V). Hence, H2O will get preference to get reduced at the cathode and as a result, H2 is evolved.
NaCl(aq)Na+(aq) + Cl-(aq)
At cathode: 2H2O(l) + 2e- H2(g) + 2OH-(aq)
At anode: Cl-(melt) Cl(g) + e-
2Cl(g) Cl2(g)
25. What is the role of graphite rod in the electrometallurgy of aluminium?
Ans. In the electro metallurgy of aluminium, a fused mixture of purified alumina Al2O3, cryolite(Na3AlF6) and fluorspar (CaF2) is electrolysed. In this electrolysis, graphite is used as the anode and graphite-lined iron is used as the cathode.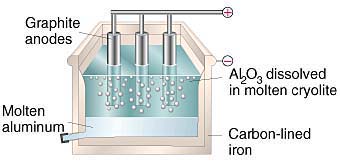
During the electrolysis, Al is liberated at the cathode, while CO and CO2 are liberated at the anode, according to the following equation.
Cathode: Al3+(melt) + 3e- Al(l)
Anode: C(s) + O2+(melt) CO(g) + 2e-
C(s) + 2O2-(melt) CO2(g) + 4e-
If a metal is used instead of graphite as the anode, then O2 will be liberated. This will not only oxidise the metal of the electrode, but also convert some of the Al liberated at the cathode back into Al2O3. Hence, graphite is used for preventing the formation of O2 at the anode. Moreover, graphite is cheaper than other metals.
26. Outline the principles of refining of metals by the following methods:
(i) Zone refining
(ii) Electrolytic refining
(iii) Vapour phase refining
Ans. (i) Zone refining: This method is based on the principle that impurities are more soluble in the molten state of metal (the melt) than in the solid state. In the process of zone refining, a circular mobile heater is fixed at one end of a rod of impure metal. As the heater moves, the molten zone of the rod also moves along with it. As a result, pure metal crystallizes out of the melt and the impurities pass to the adjacent molten zone. This process is repeated several times, which leads to the segregation of impurities at one end of the rod. Then, the end with the impurities is cut off. Silicon, boron, gallium, indium etc. can be purified by this process.
(ii) Electrolytic refining: Electrolytic refining is the process of refining impure metals by using electricity. In this process, impure metal is made the anode and a strip of pure metal is made the cathode. A solution of a soluble salt of the same metal is taken as the electrolyte. When an electric current is passed, metal ions from the electrolyte are deposited at the cathode as pure metal and the impure metal from the anode dissolves into the electrolyte in the form of ions. The impurities present in the impure metal gets collected below the anode. This is known as anode mud.
Anode: M Mn+ + ne-
Cathode:Mn+ + ne- M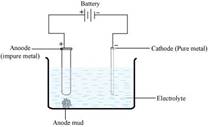
(iii) Vapour phase refining: In this method , impure metal is first converted into a volatile compound by heating with a chemical reagent at lower temperature.After this, the volatile compound is decomposed by heating to give pure metal.. e.g. Mond's process for the extraction of nickel and Van Arkel method for preparing ultra pure titanium are based on the principle of vapour phase refining.
27. Predict conditions under which Al might be expected to reduce MgO.
Ans. The equation for the formation of the two oxides are:
If we look at the plots for the formation of the two oxides on the Ellingham diagram, we find that the value of Al2O3, at temperatures below 1623K is less negative than that of MgO.
Thus, of the reaction: is negative. Therefore, below 1623K, Mg can reduce Al2O3 to Al. However above 1623K, the value for Al2O3 is more negative than that of MgO. Thus, of the reaction is negative. Therefore, above 1623K, Al can reduce MgO to Mg.